 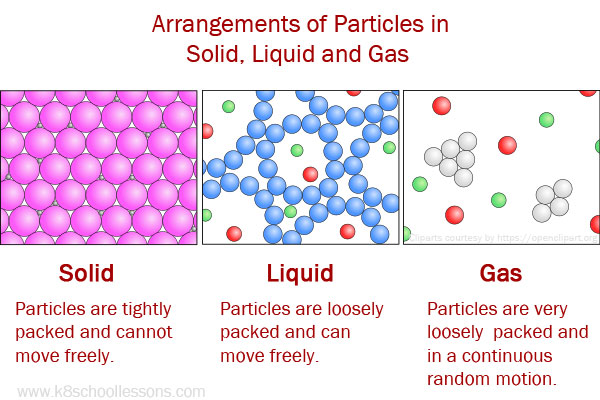
Diagram showing heat required to heat water from solid ice to liquid water and to vapor (steam). The amount of energy required is call the latent heat of freezing or boiling.įigure 7.12. Likewise, energy must be withdrawn to change the state when cooling the material. The reason is that energy must be expended to change the state from solid to liquid or from liquid to gas. The same thing applies when cooling the materials. That means ice water will remain at 0° C ( 32° F) until all the ice is melted. Cooling the bulk solid is often required for downstream processing or handling. High-temperature bulk solid streams often need to be cooled, notably in industries such as minerals processing, carbon/ash and other specialty products. After learning about changing states of matter, and conversion from solid to liquid, let us now have a look at the melting point. Solids to Liquid High-temperature heat transfer solutions. When any material is heated to the temperature where it changes state, the temperature will remain the same until all the material changes state. The process of obtaining solid from the liquid is the opposite remove heat from the sample and it is called fusion or freezing. To convert 1 gram of water at 100° C to 1 gram of steam at 100° C requires 540 calories. To convert 1 gram of ice at 0° C to 1 gram of water at 0° C requires 80 calories. As water cools it will reach 32☏ (or 0° C) is will stay at that temperature until all the water freezes (Figure 7.12). For instance a pot filled with water on the stove will gradually warm up until the water temperature approaches 212° F (or 100° C)-it will stay at that temperature until all the water has boiled away. Latent heat is the heat required (measured in calories burned) to convert a solid into a liquid or vapor, or a liquid into a vapor, without a change of temperature. Because it takes more energy to convert a substance from one physical state to another (solid to liquid, or liquid to gas), those transitions require a larger amount of energy.  
When any material is heated to the temperature where it changes state (converting from solid to liquid, or liquid to gas), the temperature will remain the same until all the material changes state. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
